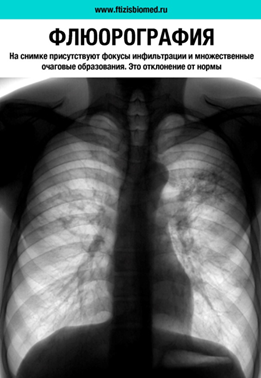
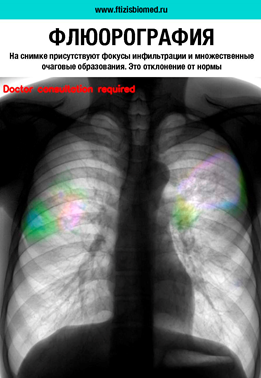
Technical trials of a medical device «Program for automated analysis of digital fluorograms»
In the period from February 2020. until April 2020. «Program for automated analysis of digital fluorograms» LLC «FtizisBioMed» passed technical tests at the testing center of LLC «Medico-Technological Agency» in Moscow. The trials included a study of the program,its evaluation and analysis data of medical device. The purpose of these trials was to determine matching the characteristics of a medical device meet the requirements of regulatory documentation.
Trials were carried out for compliance with the requirements:
— GOST 20790-93 «Instruments, apparatus and equipment for medical. General specifications» — GOST 28195-89 «Evaluation of quality of software tools. Generalities» — GOST P 51188-98 «Information protection. Trials of software tools for the presence of computer viruses. Standart guide» — GOST Р ISO 9127-94 «Information processing system. User documentation and packaging information for consumer software packages» — GOST Р IEC 62304-2013 «Medical devices. Software. Life cycle processes.» — GOST Р ISO 15223-1-2014 «Medical devices. Symbols, used for labeling on medical devices, labels and accompanying documentation. Part 1. Basic requirement»As a result of the trials, it was concluded that «The program for automated analysis of digital fluorograms» has passed technical trials, no comments on the operational and technical documentation were found. Product LLC «FtizisBioMed» has successfully passed technical trials and received an opinion on compliance with the specified requirements of regulatory documentation.
LLC «FtizisBioMed»Subsidiary enterprise JSC RC «Vector», resident of the Biomedical cluster of the Innovation center (Foundation) «Skolkovo» with the project «Information and measurement system for automated detection of diseases of organs the chest on the basis of a mobile fluorographic complex».